When building early-stage clinical trials, attention typically turns toward protocol design, recruitment and assay development. Pathology determines whether samples are usable, endpoints are measurable and biomarkers are real and yet is often treated as a supporting act.
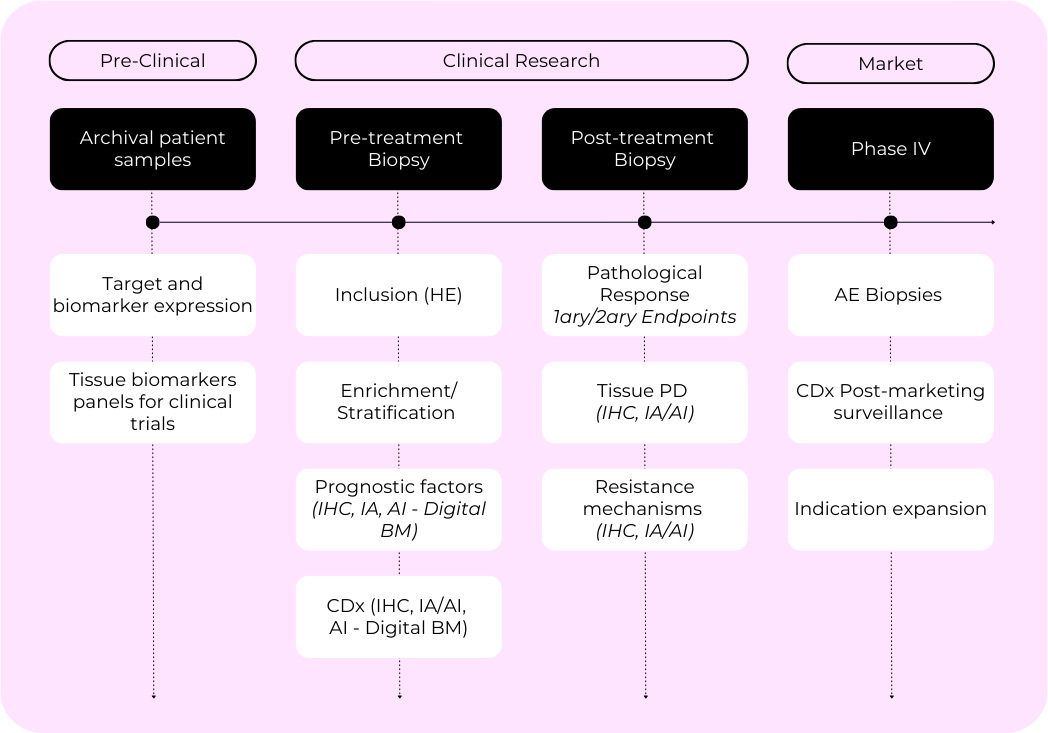
Pathology across the drug development continuum. Pathology informs decisions from early discovery through to clinical implementation, shaping how therapies are understood, validated, and monitored. Its continuous presence ensures biological relevance, consistency and translational value across every phase of development.
In reality, pathology acts as the bridge between hypotheses and clinical evidence. Every biopsy, stain, and digital image carries the proof points regulators depend on to validate your therapy. When pathology is inconsistent or under-led, that evidence weakens. When it’s expert-led and integrated early, it becomes one of the strongest assets in your clinical strategy.
At Diagnexia Analytix, we see this play out daily. Sponsors who embedded pathology expertise from the outset tended to resolve feasibility challenges sooner, achieve cleaner datasets and reach regulatory milestones faster. In our view, pathology isn’t simply an operational component. It’s a strategic discipline that determines how confidently you can stand behind your evidence.
Turning Complexity into Clarity
Early-stage and translational trials are data-dense and high-risk. You’re balancing multiple endpoints, limited tissue availability and often novel biomarkers. Without pathology expertise embedded from the start, sponsors face familiar challenges: inconsistent scoring, uncertain assay feasibility and unexpected delays once the protocol is live.
Having access to an expert pathology partner changes that equation. A good partner helps you design endpoints that are biologically sound, operationally feasible and regulator-ready. They don’t just “read slides”, they translate scientific intent into clear, measurable outcomes, ensuring your tissue strategy aligns with your trial’s clinical questions.
Together, we turn a technical function into a scientific foundation, defining what success looks like and building the data to prove it.
Building with Expertise, Not Assumptions
No two studies are alike. A pathology framework for neoadjuvant treatment in melanoma looks nothing like one for autoimmune disease or the search for new biomarkers in solid tumor. That’s why access to subspecialist pathology expertise is critical.
The right pathology partner provides named leadership, not a revolving roster. At Diagnexia Analytix, every study is guided by a Pathology Lead: a senior, board-certified specialist accountable for endpoint definition, quality control and communication between sponsor, CRO and lab.
That structure turns pathology into a literal extension of your scientific team, ensuring decisions are made with authority and traceability, not delegated down the chain.
Translating Your Data into Action
A pathology partner’s greatest value often appears before a study even starts. In pre-protocol phases, expert input ensures your biomarker plans and tissue handling requirements are aligned with both feasibility and regulatory standards.
Through early feasibility workshops and charter development, Diagnexia Analytix helps sponsors move from “What do we need to measure?” to “How do we measure it reliably, globally, and at scale?”
The outcome is a trial that runs cleaner, with fewer amendments and faster site readiness, because complexity was resolved upfront, not midstream.
Speed, Without Compromise
The demand for rapid turnaround has never been higher. Sponsors need faster reads, interim snapshots and adaptive design flexibility, but in reality, cutting corners on the quality of pathology data from slide image to diagnosis introduces silent risk: errors that surface only when data reach review boards.

The right partner combines digital workflows and scientific oversight to deliver both speed and confidence. Diagnexia Analytix’ digital-first infrastructure allows <48-hour turnaround capacity while maintaining audit trails, version control and consensus diagnoses, or adjudication for discordant cases.
In other words, you don’t have to choose between timely results and trustworthy data. You can have both.
Flexibility That Scales With You
Your needs at proof-of-concept aren’t the same as at pivotal phases of development. That’s why a flexible, modular engagement model matters. You may need a one-off endpoint feasibility review or a fully embedded pathology leadership function and your partner should be able to scale up or down without losing continuity.
Diagnexia Analytix’s approach is built on this principle: supporting sponsors with customisable engagement options, from early input and charter authorship to full global QC oversight and consensus review. It’s a partnership designed to grow and scale as your pipeline does.
Embedded Leadership: Pathology as Part of Your Team
In high-stakes research, it’s often difficult to trust third-party providers with your pathology data and workflows. It can feel like a risk that has the potential to introduce disconnect and blur outcomes. In an ideal environment, pathology ‘outsourcing’ has to be embedded and connected to your trial operations, clinical leads and biomarker scientists in real time. Instead of a disconnected third-party that covers monotonous tasks, it’s possible to embed pathology leadership directly into your team to take responsibility, managing and communicating outcomes.
An embedded pathology lead does not only provide clinical and scientific expertise, but ensures continuous alignment across CROs, labs and sponsor teams. They manage escalations. They chair technical calls and oversee data quality so issues are solved before they become delays. They become an extension of your research organisation and your priorities become their priorities.
This is what differentiates a vendor from a partner. One executes tasks; the other owns the scientific outcome alongside you.
The Diagnexia Analytix Model: How it Works
Diagnexia Analytix was built around a simple idea that pathology should enable precision medicine, not limit it. Our model combines:
- Leadership: Named Pathology Leads accountable for scientific and operational success.
- Frameworks: Charters, SOPs and QC plans aligned with GCP, ISO and regulatory expectations.
- Digital Workflows: Whole-slide imaging, automated QC and multi-reader adjudication.
- Integration: Pathology linked with genomics, spatial biology and clinical outcomes for translational insight.

The Impact
When pathology is treated as a strategic discipline, sponsors achieve more than efficiency.
They gain:
- Faster study start-up and smoother global alignment
- Reproducible, auditable data packages for submission
- Stronger regulator confidence in biomarker endpoints
- A true partnership that scales across their development portfolio
In Summary
When you outsource Diagnexia Analytix, you’re not outsourcing a task, you’re choosing a partner to help shape the pathology chapters in the story of your trial.
A great pathology partner doesn’t just process samples. They turn biology into evidence. They help you see around corners, maintain quality under pressure and build your precision medicine strategy with total confidence.
Diagnexia Analytix: Expert-led. Digitally enabled.
Let’s define your pathology strategy early.

.svg)

